I. Non-Destructive Lung Tissue Stiffness Quantification
In living tissues, mechanical stiffness and biological function are intrinsically linked. Alterations in the stiffness of tissues can induce pathological interactions that affect cellular activities and tissue function. Accurate measurement of lung tissue stiffness has been challenging due to the anatomical and mechanobiological complexities of the lung. Discrepancies between measured stiffness of dissected lung tissue samples and intact lung have limited the ability to accurately characterize integral lung stiffness. Aiming to address this challenge, we are developing a non-destructive vacuum-assisted method that enabled accurate determination of the mechanical properties of lung tissues.

II. Functional Regeneration of Refused Donor Lungs
Lung transplant, the only clinical option available for patients with end-stage lung disease, is often delayed due to the severe shortage of viable donor lungs. While timely organ transplantation is one of the major contributors to favorable treatment outcomes, more than 80% of donor lungs are rejected for transplant. Through collaboration with a research team at Columbia University Medical Center and University of Southern California, we are pursuing a stem cell-based lung regeneration approach where damaged or injured airway epithelium of donated lungs is replaced with healthy epithelium to promote lung function recovery.

III. Targeted Respiratory Drug Delivery
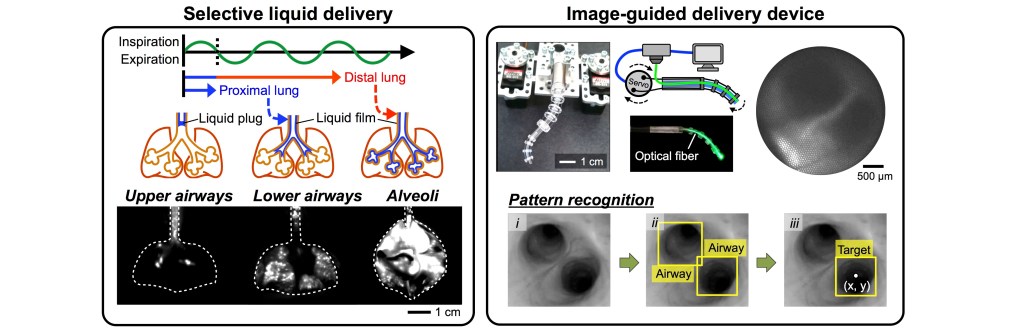
Systemic drug administrations suffer from inefficient delivery to a specific pathologic region of the lung. Our group is exploring an alternative method for delivering drugs directly to specific lung regions in the form of liquid microvolumes. In addition, we develop fluorescent imaging devices and systems that allow real-time in situ monitoring of the lung. We envision that our method would enable predictable drug concentrations at the target site, reduce the amount of the drug required for treatment, and minimize systemic side effects for several lung diseases such as lung infection and cancer.
IV. Microfluidic Devices for Rapid Disease Diagnosis
Respiratory infections such as pneumonia are the leading cause of global morbidity and mortality. While early identification of the responsible pathogens is critical to choose optimal patient-specific antimicrobial therapy, existing detection methods are ineffective in producing clinically meaningful diagnostic results in a timely manner. To address this limitation, our group is developing minimally invasive optically guided technologies that can allow rapid identification of pathogens in local airways utilizing micro-optical imaging technique and fluorescently engineered molecular reporters such as aptamers.
